 Figure 1
Figure 1
Glaucoma is a world-leading cause of blindness. The major risk factor for glaucoma is elevated intraocular pressure (IOP), with the sole treatment for glaucoma patients being to reduce the IOP.
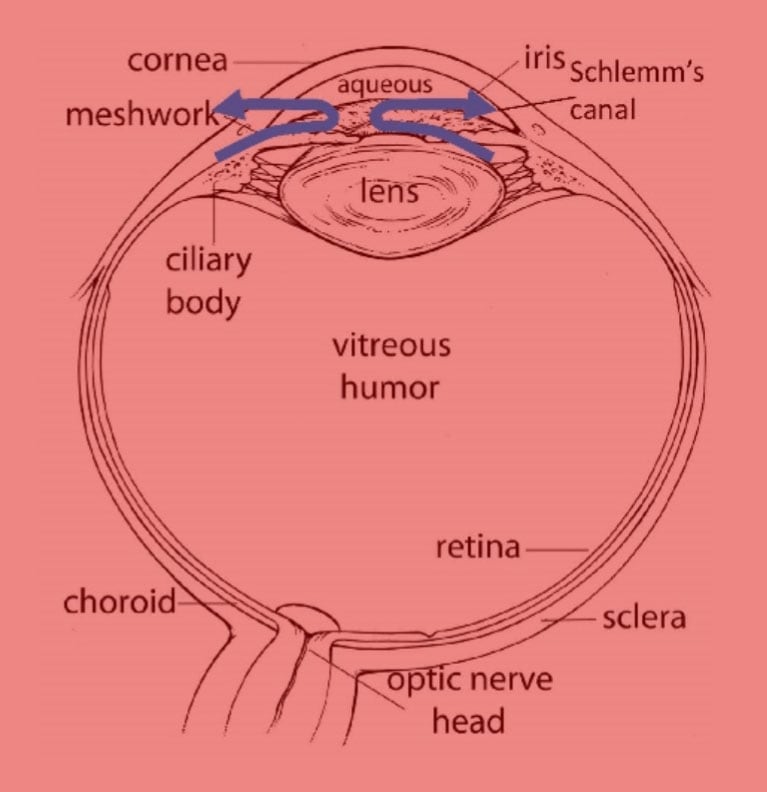
Intraocular pressure is regulated by the balance between the secretion of aqueous humour (AH) and its flow across the conventional outflow pathway, comprising the resistance of the trabecular meshwork and Schlemm's canal, a vessel that encircles the front of the eye (see Figure 1). Increased resistance of this conventional outflow pathway is largely responsible for the elevated IOP observed in glaucoma patients.
Research is now focusing on developing treatments that target the root cause of the increased resistance across the conventional flow pathway, which is characterized by the outflow facility (the reciprocal of resistance). This requires the ability to measure flow and pressure during ocular perfusion. Ocular tissue is too complex to accurately mimic with in vitro methods (e.g. organ-on-a-chip). Mouse eyes, which are anatomically similar to human eyes, are used in this research. However, their small size requires extremely high measurement precision. The use of animal tissue for the research makes it critical that the measurements are optimized to maximize the information extracted.
Conventional measurement approaches have been shown to introduce considerable errors in estimating the outflow facility (up to >250% error). Furthermore, existing measurement methods were far from robust, a difficulty exacerbated by the fact that the users of such systems are experts in biology, pharmacology, and physiology, but are typically not engineers.
To overcome these issues, the iPerfusionTM system, designed by Dr. Joseph M. Sherwood, Faculty of Engineering, Department of Bioengineering at Imperial College London, offers a new approach to measure and analyze outflow facility. This system uses an automated hydrostatic pressure reservoir to control pressure and a thermal flow meter to measure the flow rate.
Products Used
Omega PXM409: High accuracy wet-wet differential pressure transducer with 5-point NIST traceable calibration provides high stability, low drift, and premium temperature performance across a broad compensated temperature range. This pressure transducer is ruggedized with secondary containment and welded stainless steel construction and can be customized. The high accuracy and stability of this sensor is critical for the precise measurements required in iPerfusion.
Omega TXDIN1620: A new generation DIN rail mounted temperature transmitter, the TXDIN1620 is designed to accept the most common process and temperature sensor inputs and provide users with a standard two-wire 4 to 20 mA output signal. In this application, the TX DIN was used to convert the RTD signal to a 0-10v output. Isolation is provided between input and output, and all temperature ranges are linear to temperature. It is designed for ease of use with a USB interface and quick and easy configuration.
Omega Nozzle Band Heaters: This reliable mica insulated band heater was used to wrap around the bath with the temperature measured using the surface mount RTD. This band heater provides good life efficiency and dependability. It assures maintaining the lowest winding temperatures possible, keeping a low-mass heating element assembly for fast heat-up and quick thermal response to controls. It also incorporates the low thermal expansion built-in strap, a unique design feature, and can perform at higher operating temperatures [up to 480°C (900°F)]. These heaters are widely used on operations involving heating of cylindrical surfaces and are manufactured in a full range of standard construction variations, physical dimensions, electrical ratings, and a complete arrangement of screw terminals and lead terminations.
Omega SA2 Series RTD: These platinum resistance temperature sensors (RTDs) are available in 2 different mounting styles for flat or curved surfaces with a temperature range from -30 to 150ºC. The RTDs are self-adhesive for easier mounting and faster response times.
How it Works
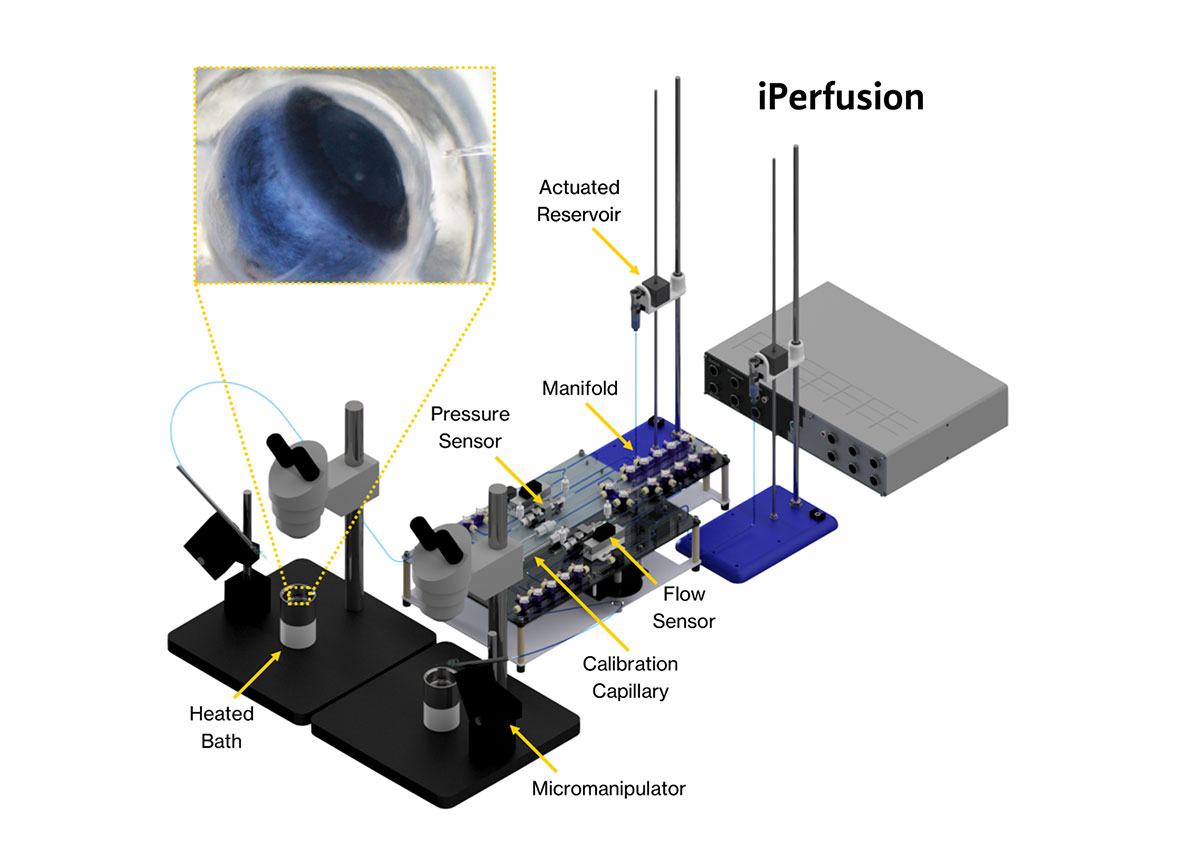
- The hardware of the iPerfusion application consists of four main components: a thermal flow sensor, a differential pressure transducer, an actuated reservoir, and a custom machined saline bath heated with a nozzle heater with PID control feedback from a surface mount RTD temperature sensor.
- To control the pressure applied to the system, a 10 ml reservoir is filled with water and coupled to a vertically mounted linear actuator with a step size of 1.25μm.
- The fluid passes from the actuated reservoir through the flow sensor (Sensirion SLG150), which has two temperature sensing elements bonded on either side of a heating element, from which the flow rate can be calculated with an accuracy of better than 10 nl/min.
- The pressure difference across the outflow pathway, which is the pressure inside the eye minus the pressure in the bath, is measured using a custom wet-wet differential pressure transducer (Omega PX409).
- To control the flow path and to allow switching between configurations required for sensor calibration and system validation, the system uses manual manifolds.
- The mouse eye is completely submerged in a relatively large volume of phosphate-buffered saline, which is regulated at 35±0.5 °C using the nozzle heater.
- The temperature of the bath is measured with a temperature sensor (Omega SA2C SA2F-RTD). The TXDIN 1620 is used to convert the signal from the RTD to a voltage output.
- Custom-built software and electronics hardware are used to control and measure the parameters of interest, providing an automated stepping procedure to acquire the flow-pressure relationship for a given eye, from which outflow facility can be calculated.
- Two mirrored systems are provided, such that one eye can act as a control, while the experimental condition is applied to the contralateral eye.
The Results
The iPerfusion system provides game-changing increases in accuracy, robustness, and speed compared to other conventional measurement approaches. As a result, it has become the leading technology for measuring outflow facility in mouse eyes, which is central to numerous novel glaucoma research projects.
Although this application focuses on enucleated mouse eyes, the system can be used to measure outflow facility in any species, and with minor modifications can be applied in vivo. The system is currently being used in 15 academic and research centers across the globe, and 21 papers have been published using the technology.
Pro Tip
Omega offers the ability to customize pressure transducers to fit the unique needs of an application. Custom configuration is available for the type of material, process fitting, pressure type, range unit, output, accuracy, electrical termination, cable length, temperature range, and more.

